Understanding Alkanes with Functional Groups
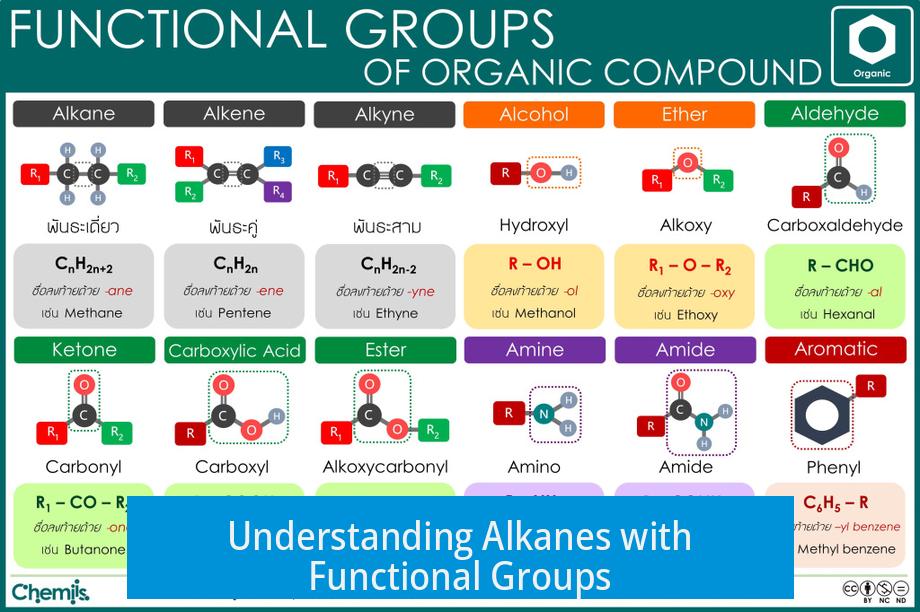
Alkanes with functional groups are organic compounds derived from alkanes but contain additional atoms or groups that modify their chemical behavior and naming conventions. These functional groups affect how the molecule is named, especially the suffix used in the International Union of Pure and Applied Chemistry (IUPAC) nomenclature.
What Defines an Alkane?
Alkanes consist only of saturated carbon-carbon (C-C) and carbon-hydrogen (C-H) bonds. Their general formula is CnH2n+2. The suffix ‘-ane’ indicates these saturated hydrocarbons.
The Impact of Functional Groups on Naming
Functional groups are specific atoms or groups of atoms that replace hydrogens in alkanes, changing their chemical properties and names.
- Groups like hydroxyl (-OH) convert the suffix from ‘-ane’ to ‘-ol’, making the compound an alcohol.
- Others, such as halogens (e.g., Br, Cl), are treated as substituents rather than functional groups that change the suffix.
Halogens: Substituents, Not Functional Suffix Modifiers
When a halogen atom replaces a hydrogen in an alkane, the compound is called a haloalkane. Despite containing additional atoms, haloalkanes retain the ‘-ane’ suffix.
For example, bromomethane (CH3Br) is a haloalkane but still ends with ‘-ane’ because the halogen only acts as a substituent. It does not qualify as a functional group that changes the suffix.
The Misconception About the ‘-ane’ Suffix
Not every compound ending in ‘-ane’ is a pure alkane. Some derivatives, including haloalkanes, phosphane (PH3), and dioxane (an ether), also end with ‘-ane’ but possess distinct functional characteristics.
| Compound Type | Example | Suffix | Effect on ‘-ane’ |
|---|---|---|---|
| Alkane | Methane | -ane | Base saturated hydrocarbon |
| Haloalkane | Bromomethane | -ane | Halogen substituent, suffix unchanged |
| Alcohol | Ethanol | -ol | Hydroxyl functional group, suffix changes |
| Phosphane | PH3 | -ane | Contains phosphorus, not an alkane |
| Ether | Dioxane | -ane | Ring ether, suffix retained |
Summary of Key Points
- Alkanes contain only saturated C-C and C-H bonds and have the suffix ‘-ane’.
- Functional groups like hydroxyl (-OH) change the suffix to indicate a different compound class.
- Halogens are substituents in haloalkanes and do not alter the suffix ‘-ane’.
- Not all compounds ending in ‘-ane’ are alkanes; some are derivatives or contain other heteroatoms.
What defines a functional group that changes the suffix in alkanes?
Functional groups that change the suffix are groups like OH, which changes the suffix to -ol. They alter the chemical name by indicating specific reactive sites.
Do halogens like Br and Cl affect the suffix in alkane naming?
Halogens act as substituents. They do not change the suffix of the alkane but are named as prefixes, making the compound a haloalkane.
Are all compounds ending with -ane simple alkanes?
No, although all alkanes end with -ane, many compounds like haloalkanes, phosphane, and dioxane also end in -ane but are not true alkanes.
What makes a haloalkane different from a regular alkane?
Haloalkanes are alkane derivatives containing halogens. The presence of a halogen replaces a hydrogen atom but the compound still keeps alkane-like features.
Can you give examples of compounds ending in -ane that are not alkanes?
Examples include phosphane (PH3), haloalkanes, and dioxane (an ether). These share the suffix but differ in structure and reactivity from simple alkanes.




Leave a Comment