Why Is the Reaction of Br2/H2O Called Halohydrin Formation?
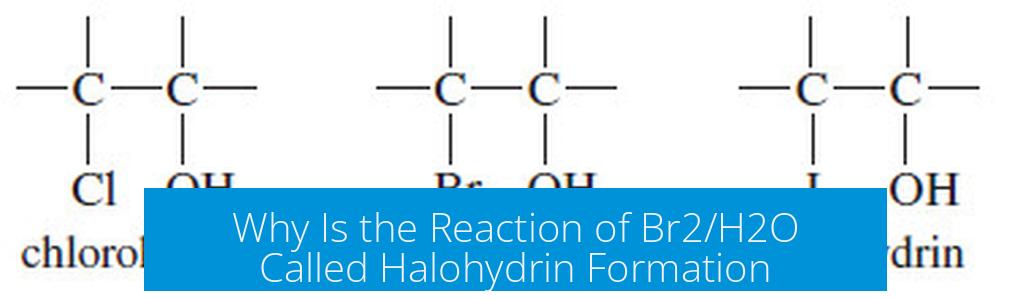
The reaction of an alkene with Br2 in aqueous solution (Br2/H2O) produces a halohydrin, where a bromine atom and a hydroxyl group add across the double bond in an anti fashion, with the OH attaching to the more substituted carbon. This reaction differs from bromination in non-aqueous solvents, which produces vicinal dibromides instead.
Understanding Halohydrin Formation
When an alkene reacts with bromine (Br2) in the presence of water, the halogen and hydroxyl groups add across the double bond. This process is called halohydrin formation. The key features include:
- Regioselectivity: The OH group bonds to the most substituted carbon of the double bond.
- Stereochemistry: The Br and OH add in an anti arrangement, forming trans stereoisomers.
- Product formation: Both enantiomers of the halohydrin often form in roughly equal amounts.
This occurs because the Br2 first forms a bromonium ion intermediate. Water then attacks the more substituted carbon due to carbocation-like character, leading to the observed regiochemistry and stereochemistry.
Contrast with Other Bromination Conditions
When the same alkene reacts with Br2 in non-aqueous solvents such as carbon tetrachloride (CCl4) or dichloromethane (CH2Cl2), the product is not a halohydrin. Instead, bromine atoms add across the double bond to give a vicinal dibromide. This results also from anti addition but lacks the hydroxyl group.
| Reagent | Major Product | Key Features |
|---|---|---|
| Br2 / H2O | Halohydrin | Br and OH; OH on more substituted carbon; anti addition |
| Br2 / CCl4 (or CH2Cl2) | Vicinal Dibromide | Two Br atoms; anti addition; no OH |
Addressing Common Confusion
Sometimes, when a question about Br2/H2O reaction is marked wrong for halohydrin formation, it may stem from misunderstanding or mislabeling the conditions.
If the product drawn has Br and OH attached correctly—with the OH on the more substituted carbon and the groups arranged anti—the answer should be considered correct.
Misgrading can occur if the reaction environment or product stereochemistry is overlooked. Accuracy requires noting aqueous solvent presence and proper regiochemistry.
Summary of Key Points
- Br2/H2O reaction forms halohydrins with Br and OH groups across the double bond.
- The OH attaches to the more substituted carbon, and addition is anti.
- In non-aqueous solvents with Br2, vicinal dibromides form instead.
- Bromonium ion intermediates explain the regio- and stereoselectivity.
- Proper recognition of reaction conditions is essential for correct answers.




Leave a Comment