Hybridization of Nitrogen Due to Resonance

Nitrogen involved in resonance is generally sp2 hybridized. This occurs because the nitrogen atom participates in pi bonding through one of its p orbitals, a hallmark feature of sp2 hybridization.
Understanding Nitrogen’s Hybridization in Resonance
When nitrogen engages in resonance structures, one lone pair can delocalize to form a pi bond with an adjacent atom. This pi bond requires a p orbital, which means nitrogen’s electron cloud rearranges itself to accommodate this participation.
- Three sp2 hybrid orbitals are formed and lie in a plane.
- The remaining unhybridized p orbital overlaps sideways to create the pi bond.
- This arrangement fits nitrogen having three regions of electron density: two sigma bonds and one pi system involving the lone pair.
This contrasts with sp3 hybridization, where nitrogen would have four regions of electron density—typically found when the lone pair remains localized and not involved in delocalization.
Common Misconceptions and Clarifications
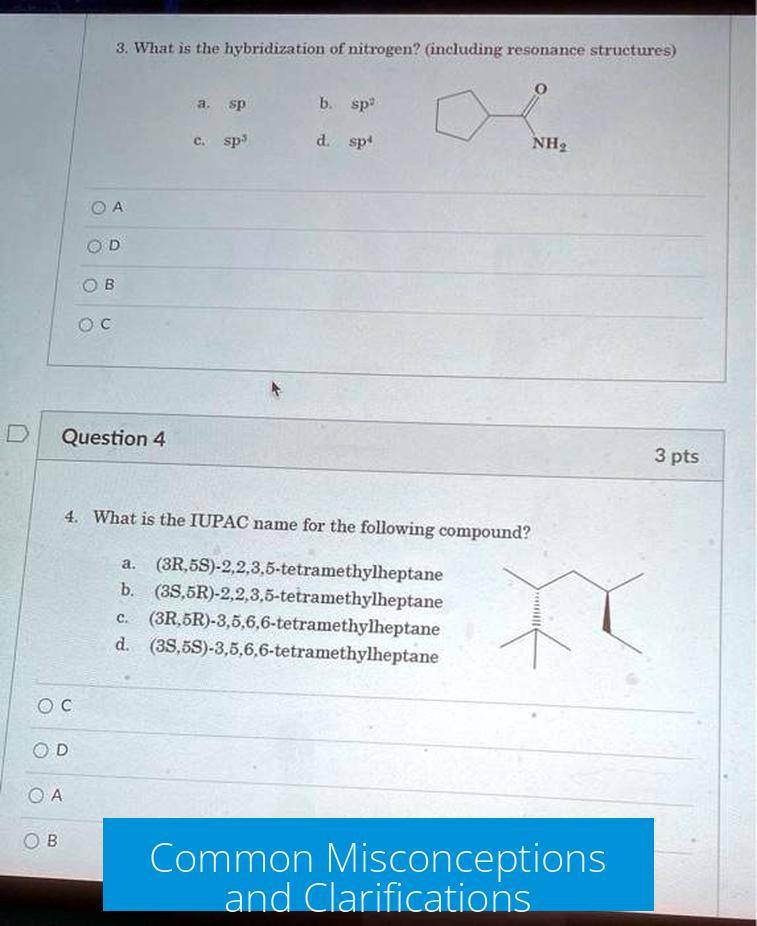
Some initial views suggested that resonance structures do not change hybridization, implying retention of sp3. However, delocalization of lone pairs in resonance affects orbital hybridization, switching coordination to sp2.
Counting electron regions alone can mislead. For example, nitrogen with two bonds to carbons, one to hydrogen, and a lone pair might appear to have four regions. Yet, if the lone pair participates in resonance, it occupies a p orbital, not a hybrid orbital.
Computational and Experimental Validation
Advanced quantum chemical calculations, such as B3LYP density functional theory methods, confirm the sp2 character of nitrogen in resonance environments.
These calculations analyze electron density distributions and validate resonance effects on hybridization more reliably than simple structural counting.
| Hybridization | Description | Relevant Electron Regions |
|---|---|---|
| sp2 | Resonance with lone pair delocalization, pi bonding | 3 (2 sigma bonds + 1 pi bond from lone pair) |
| sp3 | No resonance; lone pair localized | 4 (3 bonds + 1 lone pair) |
Key Takeaways

- Nitrogen in resonance adopts sp2 hybridization due to pi bonding via a p orbital.
- Resonance affects nitrogen’s orbital hybridization, overriding simple electron region counting.
- Advanced computational methods like B3LYP confirm this hybridization state.
What Is the Hybridization of the Nitrogen Due to Resonance?
Let’s get to the heart of the matter right away: the hybridization of nitrogen in resonance is sp2. Yes, it’s that simple—well, mostly simple. But why does falling into resonance change nitrogen’s hybridization? And what does that even mean anyway? Buckle up: we’re about to navigate the nifty world of nitrogen atoms and their orbital shenanigans.
Imagine nitrogen as a social molecule. When it gets involved in resonance, it can’t just hang out comfortably with its usual orbital setup (which many would guess as sp3, like in ammonia). Instead, it steps up its game—kind of like switching from casual jeans to a sharp blazer—to share electron density more flexibly across neighboring atoms.
Resonance Isn’t Just Party Talk — It Changes the Rules

Initially, some folks believed resonance barely touched nitrogen’s orbital hybridization. It was “just alternate bonding patterns,” they said. But science—patient as it is—showed us otherwise. When nitrogen participates in resonance, it uses a p orbital to form a pi bond. This is a game-changer.
Why? Let’s break down what this means for hybridization.
- sp3 hybridization: This happens when nitrogen has four single bonds, or three bonds plus one lone pair, using one s and three p orbitals.
- sp2 hybridization: This occurs when nitrogen is involved in one double bond (hence a pi bond) and two single bonds or lone pairs, using one s and two p orbitals for sigma bonds, leaving one p orbital free for the pi bond.
- sp hybridization: Involves a triple bond or two double bonds to nitrogen, using one s and one p orbital for sigma bonds, freeing two p orbitals for pi bonds.
When nitrogen joins resonance, that lone pair doesn’t just sit in an sp3 orbital like a couch potato. Instead, it becomes delocalized, occupying a p orbital that overlaps with neighboring atoms’ p orbitals. This creates partial double bond character. Because one p orbital is dedicated to pi bonding, nitrogen’s sigma bonding framework is better described as sp2.
Let’s Visualize the Resonance Effect
Picture a resonance structure in an amide group, for instance. Nitrogen forms a sigma bond with carbon, but the lone pair isn’t just chilling—it extends sideways in a p orbital, overlapping with a carbonyl’s p orbital. This overlap creates a partial double bond between nitrogen and carbon.
Here’s the clincher: that sideways overlap demands that nitrogen’s lone pair occupy a pure p orbital—not hybridized—while the remaining orbitals form sigma bonds via sp2 hybridization. So, nitrogen can’t be sp3 here, because having all four orbitals hybridized would leave no room for the sideways p orbital that makes resonance possible.
Hold On, What About sp or sp3? More than Just Counting Bonds
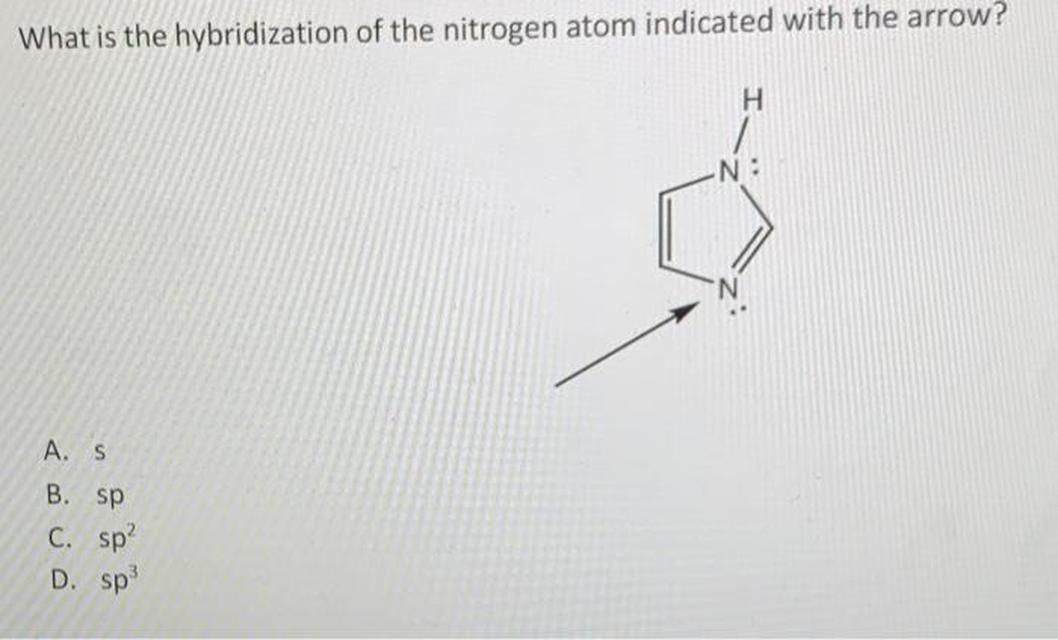
It’s tempting to say, “Hey, nitrogen has three atoms around it plus a lone pair, so that’s four regions of electron density—must be sp3!” That’s the kind of argument you might hear in an introductory chemistry class. But resonance throws a wrench in that counting method.
Whenever nitrogen participates in resonance, one of the “electron density regions” is a delocalized pi bond. Instead of treating that lone pair as localized in a hybrid orbital, the lone pair inhabits a pure p orbital overlapping adjacent atoms’ orbitals. The net result? Nitrogen effectively has three hybrid orbitals for sigma bonds (hence sp2), and one pure p orbital for pi bonding.
What about sp hybridization? Well, if nitrogen sat near a triple bond, it might be sp hybridized, but resonance in most cases involves partial double bonds (not triple bonds), so sp2 remains the more accurate assignment.
And if you want to be really, really sure—which hybridization is right—there’s good news. Computational chemists routinely run calculations (like B3LYP level theory) to analyze electron density and confirm orbital hybridizations. These calculations show that nitrogen in resonance situations behaves as an sp2 hybridized atom.
The Practical Side—Why Does This Matter?
Understanding nitrogen’s hybridization due to resonance isn’t just an academic exercise. It’s key when predicting molecular shape, reactivity, and even spectroscopy.
Here’s an example you might relate to:
- In peptide bonds: The nitrogen is part of an amide group, resonating with the adjoining carbonyl. This resonance locks the peptide bond’s planar structure and influences protein folding.
- In aromatic heterocycles: Nitrogen’s participation in resonance, adopting sp2 hybridization, affects aromaticity and how the molecule reacts chemically.
Without recognizing the sp2 nature of nitrogen in resonance, we’d misinterpret molecules’ shapes and chemical behavior. Nitrogen might look like it’s only got single bonds, but its hybridization tells the full story—namely, it’s partaking in a pi bonding party with its neighbors.
Analogy: Nitrogen’s Orbital Fashion Choices
Think of nitrogen’s hybrid orbitals as a fashion ensemble. Normally, in an sp3 setup, it wears four casual shirts—the orbitals are equivalent and symmetrical.
When resonance kicks in, nitrogen changes one of these shirts to a sleek p orbital cape that drapes sideways, overlapping with the neighbors’ capes (orbitals). This unique outfit allows resonance—delocalization of electrons—which stabilizes the molecule.
This subtle yet stylish switch from sp3 to sp2 hybridization showcases how molecular orbitals aren’t rigid but responsive to the molecule’s electronic environment.
Summary: Not Just Counting Electrons—Look for the Pi Bonds!
To sum up with confidence:
- Nitrogen in resonance adopts a strong sp2 hybridization, indicated by the presence of a pi bond and the involvement of a p orbital for the lone pair.
- Resonance affects hybridization by delocalizing the lone pair, removing it from localized sp3 orbitals.
- Computational methods like B3LYP calculations confirm this hybridization assignment.
- Simply counting bonds and lone pairs can lead you astray; watching for resonance and pi interactions is key.
Next time you spot nitrogen in a resonance structure, ask yourself: is there a pi bond involving nitrogen? If yes, coat your nitrogen in an sp2 blazer—it’s ready to mingle and resonate!
What hybridization does nitrogen have when involved in resonance?
Nitrogen is generally sp2 hybridized in resonance. This is because one of its orbitals forms a pi bond, requiring a p orbital to overlap, which matches sp2 hybridization.
Why can’t nitrogen be sp3 hybridized in resonance structures?
Although nitrogen has lone pairs and single bonds suggesting sp3, resonance involves delocalizing the lone pair into a pi bond. This uses a p orbital, which changes hybridization to sp2.
How does resonance affect nitrogen’s hybridization compared to when resonance is absent?
Without resonance, nitrogen might be sp3 or sp. Resonance requires involvement in pi bonding, so nitrogen’s hybridization shifts to sp2 to accommodate the pi electron delocalization.
Can computational methods help determine nitrogen’s hybridization in resonance?
Yes, calculations like B3LYP can clarify unsure cases by analyzing electron distribution and geometry, confirming that nitrogen adopts sp2 hybridization due to resonance effects.
Is nitrogen’s hybridization influenced by nearby multiple bonds in resonance?
Yes. The presence of double or triple bonds nearby means nitrogen’s electrons involved in resonance occupy p orbitals, enforcing sp2 hybridization to stabilize the structure.




Leave a Comment