Kinetic vs Thermodynamic Control in E1 and E2 Reactions
Kinetic control refers to product distribution governed by the reaction rate, favoring the product that forms fastest. Thermodynamic control favors the most stable product, reflecting equilibrium conditions. Applying this to E1 and E2 reactions explains variations in alkene outcomes depending on reaction conditions.
Understanding Kinetic Control
Under kinetic control, the product ratio depends on which alkene forms faster. This is dictated by the activation energy of each possible product pathway. The reaction conditions typically involve lower temperatures and shorter reaction times, where equilibrium cannot be reached.
- Product formation is irreversible within the reaction timeframe.
- The major alkene is often the less substituted but more quickly formed one.
- Common in E2 reactions with strong bases and low temperatures.
Understanding Thermodynamic Control
Thermodynamic control occurs when the reaction mixture reaches equilibrium. The product ratio reflects the relative stabilities of the alkenes. More substituted, conjugated, or trans alkenes tend to dominate as they are lower in energy.
- Reaction runs longer or under higher temperature to allow equilibration.
- The major product is the more stable alkene, often more substituted.
- Typical in E1 reactions or conditions promoting carbocation rearrangement and equilibration.
Applying Controls to E1 and E2 Mechanisms
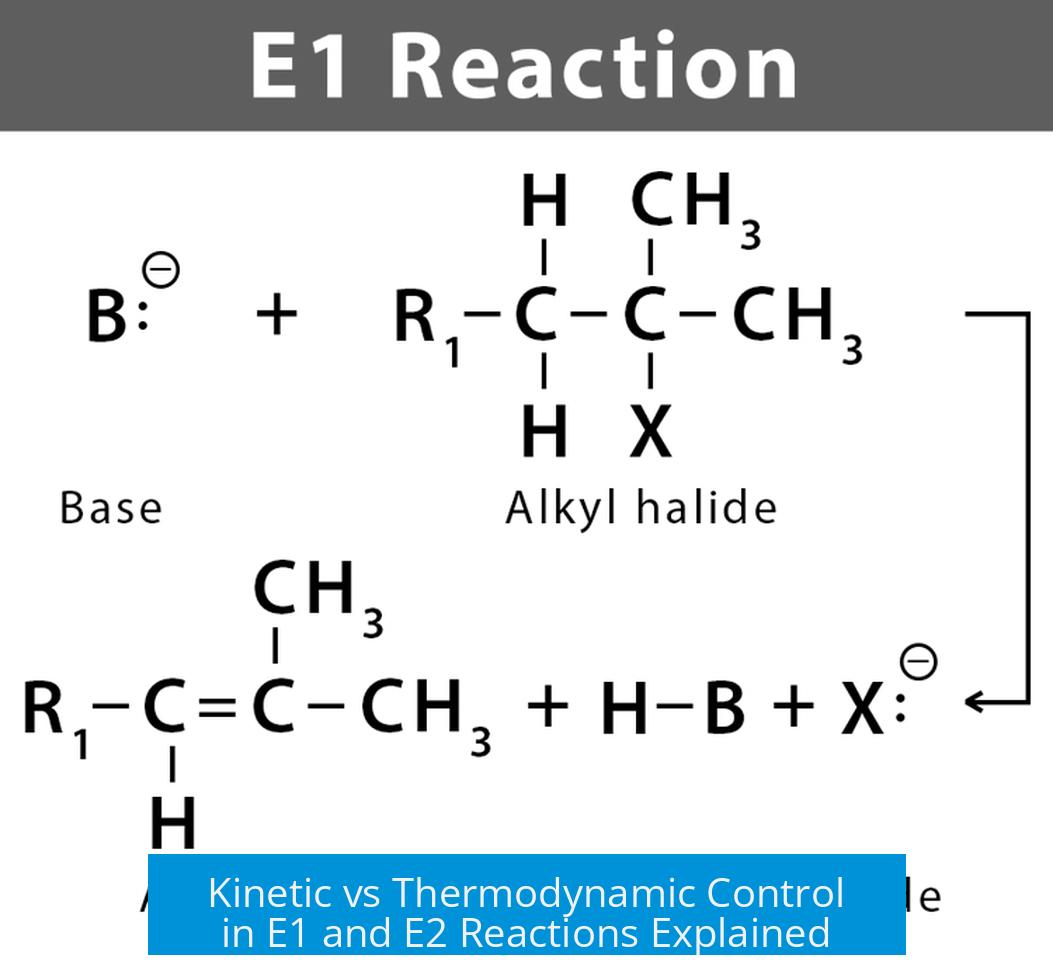
E1 reactions proceed through a carbocation intermediate, allowing rearrangement and equilibration. This typically leads to thermodynamic control, with the major product being the more stable alkene. The reaction is often under higher temperature or in protic solvents supporting carbocation formation.
In contrast, E2 reactions are concerted eliminations without a carbocation intermediate. The base abstracts a proton while the leaving group departs simultaneously. The product distribution depends on the fastest pathway, often kinetic control, favoring the alkene formed via the most accessible proton abstraction, usually less substituted.
Determining the Control Type
| Factor | Kinetic Control | Thermodynamic Control |
|---|---|---|
| Temperature | Lower | Higher |
| Reaction Time | Short | Long |
| Base Strength | Strong (favor E2) | Weaker or moderate (favor E1) |
| Solvent | Aprotic, favors elimination quickly | Protic, promotes carbocation stability |
| Product Observation | Less substituted alkene predominant | More substituted alkene predominant |
By examining these factors and the resulting alkene distribution, one can infer whether a reaction is under kinetic or thermodynamic control. For example, fast elimination with a strong base and short time likely yields kinetically controlled products. Extended heating or conditions favoring carbocations point to thermodynamic control.
Additional Considerations
- E1 reactions allow carbocation isomerization, increasing the chance for stable alkenes.
- E2 reactions require an anti-periplanar arrangement, sometimes restricting which alkene forms fastest.
- Zaitsev’s rule often applies for thermodynamic control, favoring the more substituted alkene.
- Hofmann elimination usually shows kinetic control, favoring less substituted alkenes due to steric hindrance.
Key Takeaways
- Kinetic control depends on rate and activation energy — faster forming alkene dominates.
- Thermodynamic control depends on product stability — more stable alkene dominates at equilibrium.
- E1 reactions often show thermodynamic control due to carbocation intermediates.
- E2 reactions typically exhibit kinetic control with strong bases and lower temperature.
- Reaction conditions such as temperature, base strength, solvent, and time determine the control mode.
What does kinetic control mean in the context of E1 and E2 reactions?
Kinetic control means the product formed fastest dominates. In E1/E2, this often leads to the alkene that forms more quickly, not necessarily the most stable one.
How can I tell if an E1 or E2 reaction is under thermodynamic control?
If the reaction reaches equilibrium or runs at high temperatures, the most stable alkene usually forms more. This stability-driven product distribution signals thermodynamic control.
What reaction conditions favor kinetic over thermodynamic control in elimination reactions?
Lower temperatures and short reaction times favor kinetic control. Strong bases and polar aprotic solvents also tend to speed product formation, favoring kinetically controlled alkenes.
How does the concept of activation energy relate to product distribution in E2 reactions?
The product with the lower activation energy forms faster and dominates under kinetic control. Even if it’s less stable, it appears more when the reaction is stopped early or run cold.
Can the same reaction switch between kinetic and thermodynamic control?
Yes. Changing temperature or reaction time can shift control. A reaction run longer or heated more may shift from kinetic to thermodynamic product distribution.




Leave a Comment