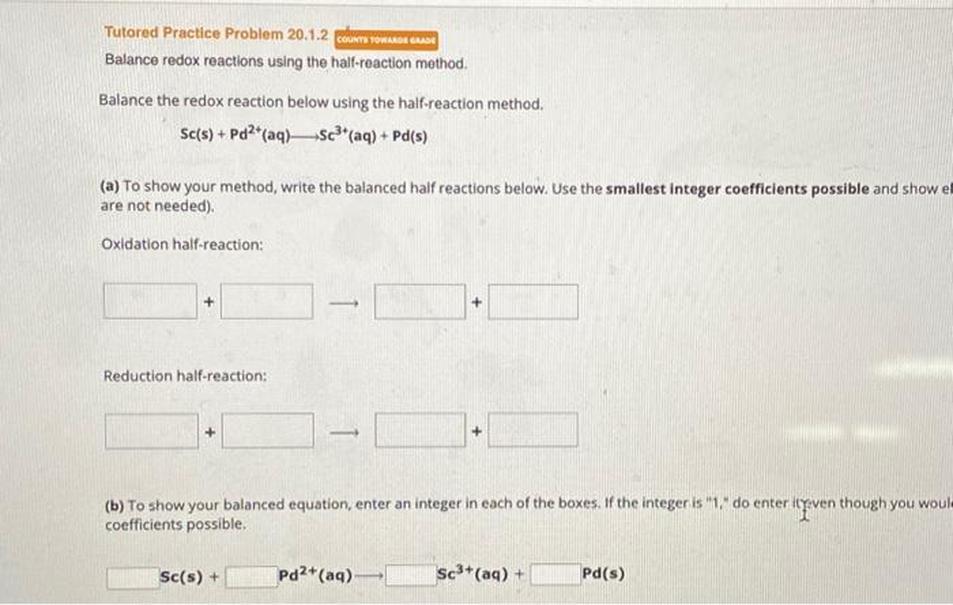
Correcting Steps to Writing a Redox Reaction with Half-Equations

Writing a balanced redox reaction using half-equations requires careful handling of electrons, species, and charge. The correct final redox equation cannot include electrons, the product must be the correct compound with accurate states, and each step must be fully balanced.
1. Electrons Must Cancel in the Final Equation
Electrons appear explicitly only in the half-equations — as oxidation and reduction steps. When combining these half-reactions, electrons must balance and cancel out so the final equation contains none.
- Do not show electrons (e−) in the final balanced redox equation.
- Presence of electrons in the final equation indicates an error combining half-reactions.
2. Use the Correct Product and Physical States
The product in many iron oxidation reactions is iron(III) oxide, Fe2O3 as a solid. Avoid showing iron ions (Fe3+) or oxide ions (O2−) as aqueous species unless the reaction physically produces them.
- Ensure charge balance by correctly representing the product (Fe2O3 solid).
- Omit water or other species if not involved in the actual reaction.
- State symbols must reflect real physical forms, e.g., (s) for solid, not (aq) when species are insoluble.
3. Correct the Oxide Formula Before Balancing
Accurate molecular formulas are essential. Using Fe2O3 rather than incorrect oxide formulas affects atom balance. Although it does not change conceptual steps, it impacts the numeric balancing of atoms and charges.
4. Intermediate Ionic Breakdown Is Acceptable But Not Final

Breaking the oxide into ionic or atomic form aids balancing. However, reassemble the oxide in the final equation to represent the true product.
- Intermediate half-equations may show ions; final equations show the compound.
- Keep the logical sequence of decomposition and recombination clear.
5. Sequence and Balancing of Steps Matter
Assigning oxidation states early is helpful, often done before balancing atoms. All intermediate steps, including Step 3, must be fully balanced in atoms and charge to avoid later inconsistencies.
- Ensure each half-equation is balanced before combining.
- Recheck the balancing at every step to ensure consistency.
- The sequence of steps can vary but must be logical, prioritizing correct oxidation state assignment and balancing.
Summary of Key Corrections
- Finalize with a balanced equation that has no electrons shown.
- Represent the product as Fe2O3 solid, not as separate ions or aqueous species.
- Break down oxides if necessary during balancing but reconstruct them in final form.
- Check all half-equations and steps are fully balanced in atoms and charge.
- Electrons in half-equations must cancel when combining; any leftover electron is a sign of error.
Correcting these steps to writing a redox reaction with half-equations
Writing a balanced redox reaction with half-equations isn’t rocket science, but it sure can feel like an episode of a reality show where electrons keep sneaking back in after they’ve been kicked out. Let’s clear up the confusion and show exactly how to write that final redox equation perfectly, with zero electrons hanging around, correct products, and proper balancing from start to finish.
So, you want to combine those half-equations and rewrite the full redox equation? Great! But hold up—before you finalize, make sure electrons completely cancel out. They should never pop up in the final answer. That’s rule number one, and if this rule sounds too strict, remember: electrons are the messengers passed between oxidation and reduction; they shuttle around during the process, but they don’t sit still at the finished reaction party.
Why Electrons Must Vanish in the Final Equation

You often see half-equations with electrons explicitly shown. For example:
4Fe → 4Fe3+ + 12e− (Oxidation) 3O2 + 12e− → 6O2− (Reduction)
When combining these, electrons must cancel out. If electrons remain in your final equation, like in this faulty example:
4Fe(s) + 3O2(g) + 6e− → 4Fe3+(aq) + 6O2−(aq),
you’ve made a mistake. Electrons appearing here scream: “You didn’t finish the job.” Either you didn’t multiply one half-equation to balance electrons or misaligned the combination. The electrons are internal carriers, not end products.
Charge Balancing: The Invisible Balancing Act
Alongside electrons, charge balancing is crucial, but it’s often overlooked. Balanced atoms? Check. Balanced charge? Sometimes not.
When iron reacts with oxygen in air, the product isn’t free-floating Fe3+ ions in solution. Nope. It’s a solid, Fe2O3, rust to all of us.
This means the final equation should respect physical states and charge neutrality:
4Fe(s) + 3O2(g) → 2Fe2O3(s).
Notice here, iron is a solid reactant, oxygen is a gas, and the product is a solid oxide. No suspicious aqueous ions or rogue electrons. The physical states matter—they tell you where species hang out and help ensure balanced equations truly represent the chemistry.
Don’t Mix up Formulae—Fe2O3 is the Star
Using the wrong oxide formula is like showing up to a costume party wearing last year’s outfit—it doesn’t ruin the logic, but you look off.
Initially, writing Fe3O2 instead of Fe2O3 might happen. It doesn’t sabotage the learning process, but mixing formulas messes with the math of balancing atoms and charge. Stick to Fe2O3 for accurate balancing.
Half-Equations: Take Your Time, But Don’t Drag Your Feet
Writing half-equations is the dance of redox chemistry. Typically, it’s best to identify oxidation states before attempting to balance the species and atoms. This approach helps clarify which elements lose electrons and which gain.
Moreover, ensure each half-equation is balanced individually:
- Atoms balanced (except for O and H if working in acidic/basic medium)
- Charge balanced (electrons added accordingly)
- States indicated (s, l, g, aq)
If you’re wobbling between balance attempts in your steps, it only means you need to pause and finalize each half-equation before combining.
The Intermediate Step: Breaking Down Oxides Carefully
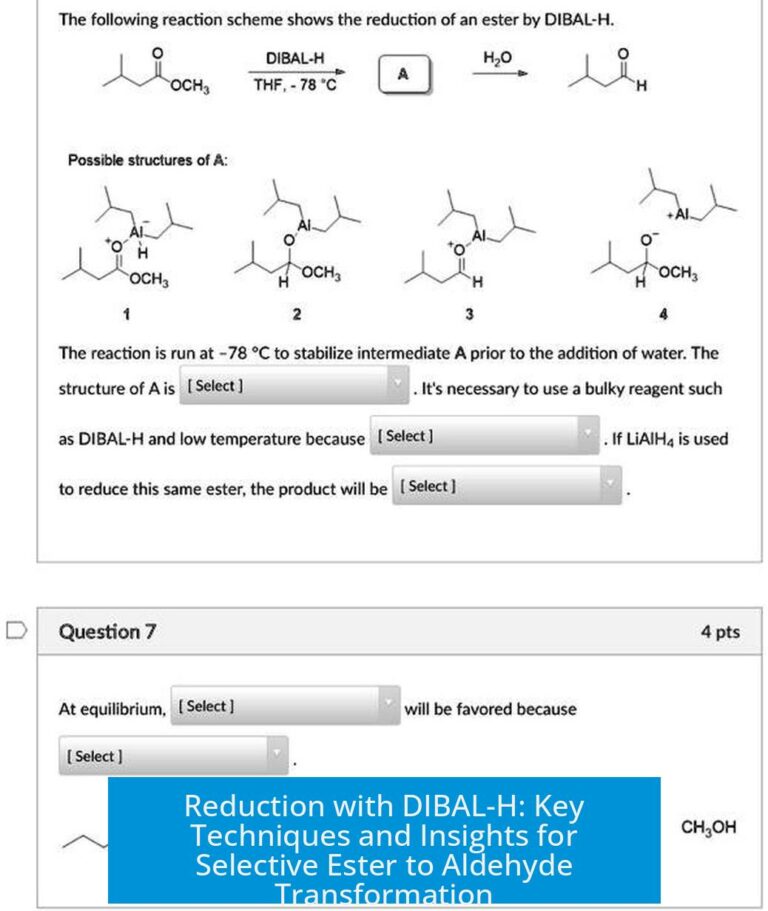
It’s perfectly fine during the process to consider the oxide breakdown as ions, like Fe3+ and O2−, to assist balancing electrons and charge. However, remember, these ionic forms are tools rather than the final destination. Your answer—especially the product—should reflect the actual compound (solid Fe2O3), not an ionic soup.
Step-by-Step Guide to Writing Balanced Redox Reactions with Half-Equations
- Identify species involved and their oxidation states. Starting with the elemental identities helps anchor your balancing efforts.
- Write two half-equations—one for oxidation, one for reduction. Balance atoms except H and O first.
- Add H2O, H+, or OH− as appropriate to balance oxygen and hydrogen. This applies especially if the reaction is in acidic or basic solutions but skip this if water isn’t involved.
- Balance charges by adding electrons. Electrons lost in oxidation equal those gained in reduction.
- Multiply half-equations to equalize electrons transferred. This is crucial to eliminating electrons when combining later.
- Add the half-equations. Confirm electrons cancel out and verify atoms and charge balance.
- Write the final equation with correct physical states. Use (s), (l), (g), (aq) correctly. No electrons or ions if they don’t exist physically.
Let’s See It in Action
Example: Iron rusting.
Oxidation (Fe to Fe3+):
4Fe(s) → 4Fe3+ + 12e−
Reduction (O2 to O2−):
3O2 + 12e− → 6O2−
Combine: electrons cancel out.
4Fe + 3O2 → 4Fe3+ + 6O2−
Translate ions into solid oxide compound:
4Fe + 3O2 → 2Fe2O3(s)
The final balanced equation has no electrons, charge is neutral, atoms balance, and states are accurate. The reaction tells the full story, clean and ready.
Wrapping It Up: Why Does This Process Matter?
Writing accurate redox reactions is more than an academic hurdle. It improves understanding of chemical changes, informs laboratory work, and prevents costly mistakes in chemistry education and industry.
A poorly balanced or improperly written redox equation can lead to misinterpretation of the reaction conditions or products. For instance, seeing Fe3+(aq) instead of Fe2O3(s) could confuse a chemist about solubility or reaction media.
Getting these steps right is like building a firm foundation before the house—skip it, and chaos follows.
Final Tips for Success
- Review each half-equation carefully before combining.
- Double-check that electrons cancel completely before finalizing.
- Confirm final product formula and physical state according to the reaction context.
- Avoid adding water or ions if they don’t belong.
- Practice with real reactions such as rusting, metal displacement, or acid-base redox to strengthen skills.
Next time you’re faced with the dreaded six electrons suddenly appearing in your final equation, take a deep breath. Step back. Balance each half-equation thoroughly and watch those electrons vanish! It’s chemistry’s version of a mic drop.




Leave a Comment