Acrylate Polymer Glue: Composition and Production
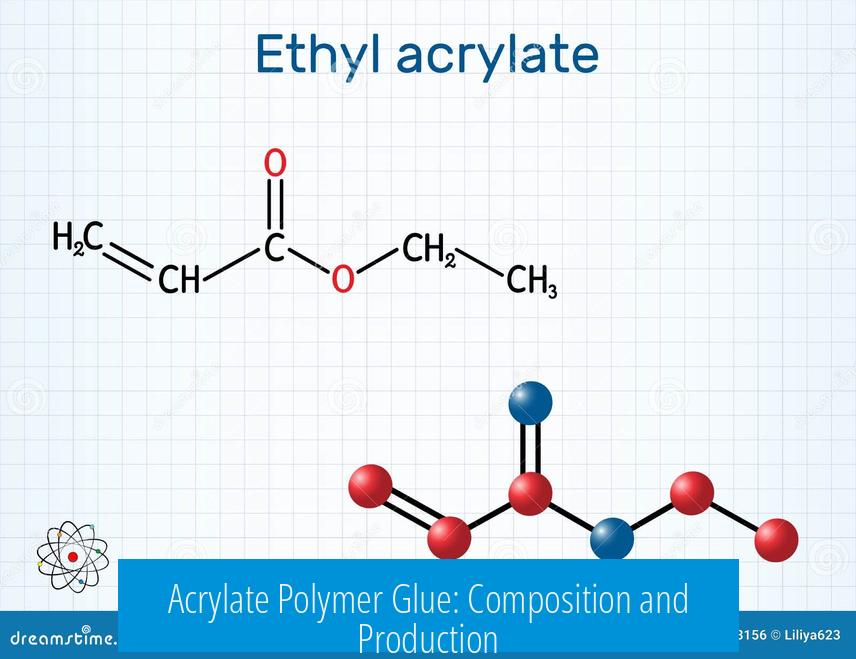
Acrylate polymer glue primarily consists of polymers formed from acrylate monomers, with methyl acrylate being a common choice for synthesis due to its well-defined properties. This polymer serves as the adhesive matrix in various bonding applications. The polymerization process, reaction conditions, and safety considerations are crucial in producing an effective acrylate-based glue.
Monomer Selection
The term “acrylate” broadly refers to a family of esters derived from acrylic acid, such as methyl acrylate, ethyl acrylate, and others.
- Methyl acrylate is commonly selected as the monomer for glue production due to its availability and suitable polymerization behavior.
- This choice provides a clear foundation for reproducible polymer synthesis and performance consistency.
Polymerization Process
Polymerization typically employs free radical initiators such as benzoyl peroxide (BPO) or azobisisobutyronitrile (AIBN).
- BPO is used in small amounts relative to the monomer (e.g., 10 mg BPO per 1 g polymer) to initiate polymerization.
- The reaction is conducted under inert atmosphere conditions (argon or nitrogen) to prevent premature termination, often using a Schlenk line or vacuum setup.
- Temperature control is critical; methyl acrylate boils near 80°C, so cooling with an ice bath is necessary during monomer handling.
- Reaction duration is approximately 24 hours to achieve satisfactory polymer conversion.
- Post-polymerization, the polymer is extracted by dissolving in a solvent like toluene and precipitating in methanol or water to purify the glue polymer.
Technical Guidance and Precautions
Manufacturing acrylate polymer glue is complex and involves risks:
- Benzoyl peroxide is explosive when heated; therefore, strict control measures are mandatory.
- Polymerization demands expertise; it is advisable to collaborate with adhesive manufacturers rather than attempting home synthesis.
- Employing trained chemists for evaluation and quality control optimizes product performance and safety.
- Researching free radical polymerization of methyl acrylate through academic sources can improve formulation accuracy and procedure refinement.
Market and Availability
Many commercial acrylate adhesives exist, offered by companies like 3M. These commercially available products provide reliable performance without the challenges of laboratory-scale synthesis.
- Purchasing established adhesives often outweighs the benefits of developing new formulations outside professional settings.
- Exploring existing product portfolios helps align adhesive selection with specific application requirements.
Reference Material
High-purity methyl acrylate monomers and related reagents are available from chemical suppliers such as Sigma-Aldrich (Methyl Acrylate Product), facilitating controlled polymer synthesis under professional conditions.
Key Takeaways
- Acrylate polymer glue is mainly synthesized from methyl acrylate monomers via free radical polymerization.
- Initiators like benzoyl peroxide enable polymer chain formation but require careful handling.
- Polymerization demands inert atmosphere, temperature control, and appropriate purification steps.
- Developing acrylate glue is technically challenging; collaboration with manufacturers and experts is recommended.
- Commercial acrylate adhesives are widely available and often preferable to homemade products.
What monomer is commonly used for acrylate polymer glue synthesis?
Methyl acrylate is typically selected as the monomer for acrylate polymer glue. It is a clear choice among acrylate compounds for this purpose.
Which initiators are recommended for polymerizing methyl acrylate?
Benzoyl peroxide (BPO) and AIBN work well as initiators. BPO needs careful handling as it can be explosive if heated. Usually, about 10 mg of BPO per gram of polymer is used.
What are the key steps in the polymerization process of acrylate glue?
- Mix BPO and methyl acrylate under inert atmosphere.
- Keep the reaction cool in an ice bath since methyl acrylate boils at 80°C.
- Use a Schlenk line or vacuum with argon/nitrogen gas.
- Allow polymerization to proceed for about 24 hours.
- Afterward, dissolve the product in toluene and precipitate it in methanol or water.
Is it advisable to make acrylate polymer glue at home or on a small scale?
No, the process is complex and requires technical expertise. It’s safer to partner with companies specializing in adhesives and use their existing products.
Where can one find commercial acrylate polymer adhesives?
Major companies like 3M offer a wide range of acrylate adhesive products. Buying these is more practical than attempting in-house development.
Are there resources available to study the polymerization of acrylates further?
Yes, academic websites and scientific articles on free radical polymerization of methyl acrylate provide detailed procedures and insights to refine the synthesis process.



Leave a Comment