Understanding Skeletal Structure in Organic Chemistry

Confusion about skeletal structures often arises from difficulty representing three-dimensional molecules accurately on a two-dimensional surface. Key principles help clarify this process and improve the accuracy of these drawings.
Maintaining Realistic Bond Angles
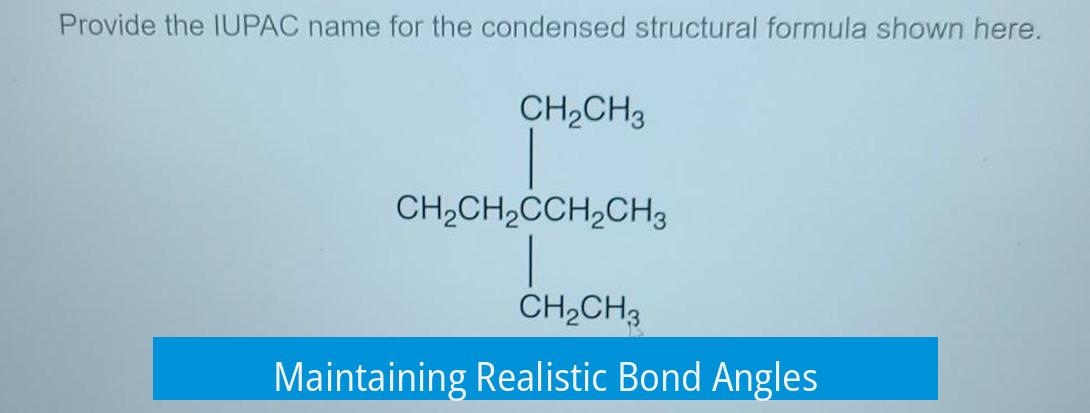
Bonds in organic molecules, especially those involving sp2 hybridized carbons, ideally measure close to 120 degrees. This angle reflects the trigonal planar geometry found in many molecules.
When drawing skeletal structures, placing atoms so bond angles approximate 120 degrees helps capture the molecule’s true shape and spatial arrangement. Attempting to reproduce this geometry makes the drawing clearer and chemically meaningful.
Drawing the Longest Chain as a Zigzag
The longest carbon chain forms the skeleton of the molecule. It is best represented as a zigzag line with alternating peaks and valleys. This geometric pattern offers a simple way to indicate the carbon backbone.
Peaks in the zigzag correspond to upward-facing carbons, while valleys point downward. Substituent groups attach accordingly: groups on peak carbons appear on top, while groups on valley carbons are shown below. This method ensures consistent and intuitive placement.
Positioning Substituent Groups to Minimize Crowding
Atoms repel each other due to steric effects. Thus, substituents should be drawn spread out evenly around the chain to reflect realistic spacing.
For example, a carbon branch next to two adjacent carbons is better placed below the chain if those neighbors are above or on peak positions. This avoids overcrowding and makes the structure easier to read.
Challenges in Visualizing Group Orientation
Determining whether groups like methyl (CH3) are “facing up” or “down” can pose difficulties without the original 3-D model or structure.
Without visual aids, describing the orientation of these groups is challenging. Using molecular models or software can assist in visualizing these orientations when needed.
Key Takeaways
- Keep bond angles near 120° to reflect real molecular geometry.
- Draw the longest carbon chain as a zigzag with peaks and valleys.
- Attach substituents on peaks (top) or valleys (bottom) for clarity.
- Position groups evenly to minimize steric crowding.
- Use visual tools to clarify difficult orientations like methyl group positions.





Leave a Comment