Covalent Bond vs Hydrogen Bond
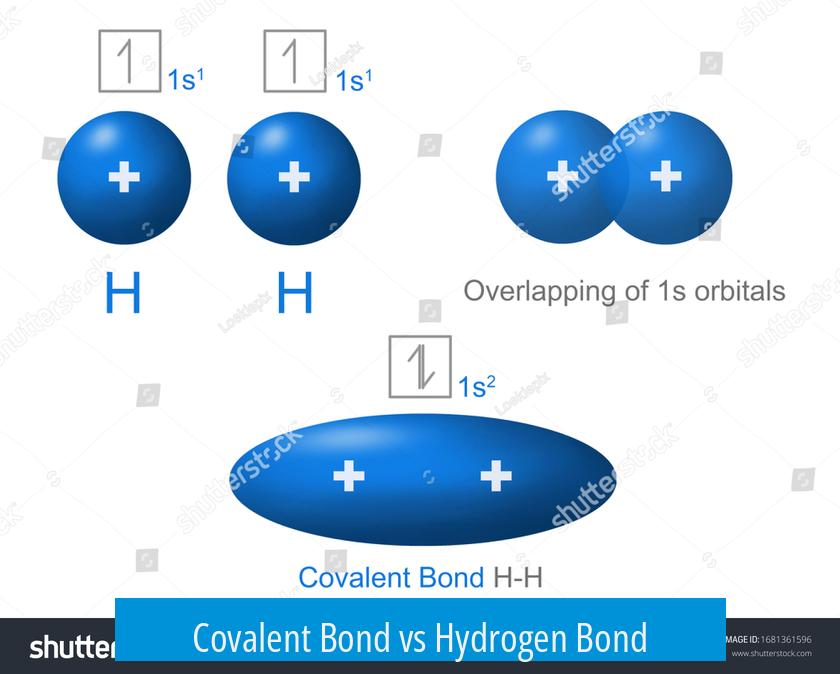
Covalent and hydrogen bonds differ fundamentally in their nature, strength, and electron behavior. Covalent bonds involve sharing electrons between atoms to form a stable molecule. Hydrogen bonds result from electrostatic attractions between polar molecules but do not involve electron sharing. Understanding these distinctions clarifies their roles in molecular structure and biological systems.
1. Nature of Covalent and Hydrogen Bonds
1.1 Covalent Bond
Covalent bonds form when two atoms share one or more pairs of electrons. This sharing occurs because atoms seek to complete their outer electron shells, usually to reach eight electrons. The bond represents a strong electrostatic attraction between the shared electrons and the nuclei of both atoms, resembling a secure, stable link.
Such bonds are powerful, providing high stability to molecules. The underlying forces follow Coulomb’s law, where the positively charged nuclei and shared electrons attract each other strongly. Covalent bonds weld atoms tightly, creating durable assemblies such as methane (CH4) or water (H2O).
1.2 Hydrogen Bond
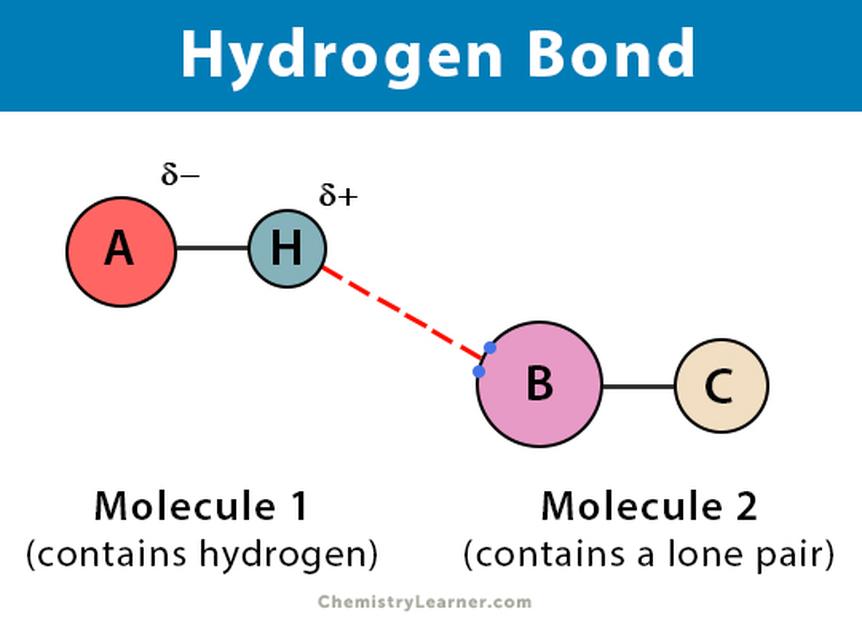
Hydrogen bonds do not involve electron sharing. Instead, they arise from the attraction between a positively polarized hydrogen atom and a negatively charged electronegative atom nearby. The interaction is purely electrostatic, akin to the attraction of opposite magnetic poles.
In these bonds, the hydrogen atom acts as a bridge connecting two molecules or different parts of a large molecule. Although weaker than covalent bonds, hydrogen bonds can exhibit some covalent-like character, representing an extreme form of dipole-dipole attraction.
2. Electron Distribution and Polarity
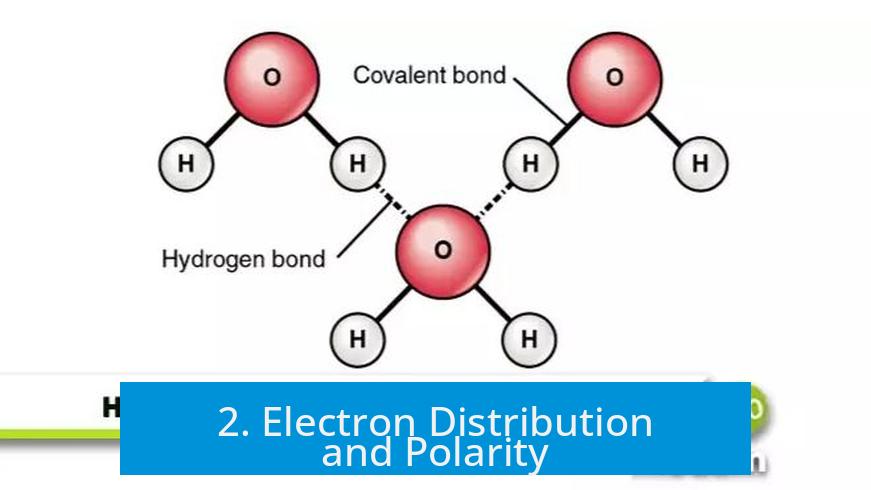
2.1 Covalent Bonds: Electron Sharing and Polarization
In polar covalent bonds like O–H, electrons spend more time near the more electronegative atom (oxygen). This unequal sharing causes partial charges, with oxygen gaining a partial negative charge and hydrogen a partial positive charge.
Each hydrogen atom can form a single covalent bond due to its single electron. The shared electrons bond the atoms tightly and define their chemical behavior.
2.2 Hydrogen Bonds: Partial Charges and Electrostatics
Hydrogen bonds emerge from the interaction between a partially positive hydrogen and a partially negative electronegative atom (often nitrogen, oxygen, or fluorine). The electron around hydrogen shifts away towards the electronegative atom, leaving hydrogen’s nucleus exposed and effectively positively charged.
This charge separation enables electrostatic attraction without electron sharing. These partial charges stabilize interactions between molecules rather than atoms within a molecule.
3. Strength and Properties of Covalent and Hydrogen Bonds
| Bond Type | Typical Strength (kJ/mol) | Effect on Molecular Stability |
|---|---|---|
| Covalent Bond | 150–1100 | Holds atoms together within molecules; resistant to heat and chemical changes |
| Hydrogen Bond | 5–40 | Stabilizes molecules interactions; impacts physical properties like boiling point |
Covalent bonds are very strong and unlikely to break under normal conditions. For example, boiling water does not break the O–H covalent bonds. Conversely, hydrogen bonds are weaker and commonly break and reform as conditions change, such as when water transitions between solid, liquid, and vapor.
4. Conceptual Analogies for Understanding
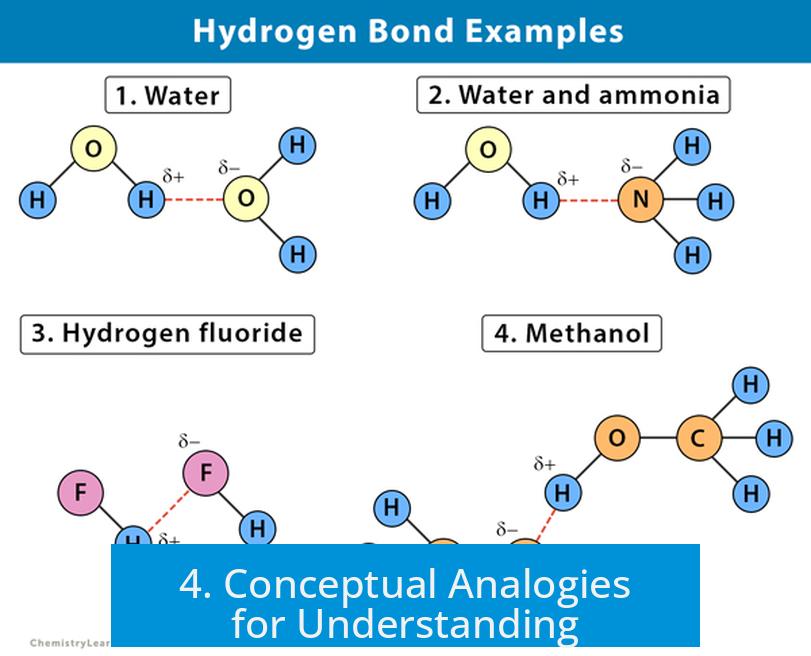
- Covalent bonds: Like permanently welding two metal parts together. They create a fixed, strong connection.
- Hydrogen bonds: Like strong magnets attracting a ferrous surface. The connection is strong but flexible and temporary.
These analogies clarify why covalent interactions define molecular identity, while hydrogen bonds influence molecular organization and behavior.
5. Continuum Between Bond Types
Bond definitions form a continuum. Hydrogen bonds are extreme dipole interactions but do not always have a sharp boundary separating them from other dipole forces. The rigidity in classifying hydrogen bonds can sometimes obscure their true nature as enhanced electrostatic interactions.
Electrons do spend some time near hydrogen in hydrogen bonds, but no actual sharing occurs as in covalent bonds. The atoms retain their electron configurations and molecular structure remains intact.
6. Biological and Molecular Examples
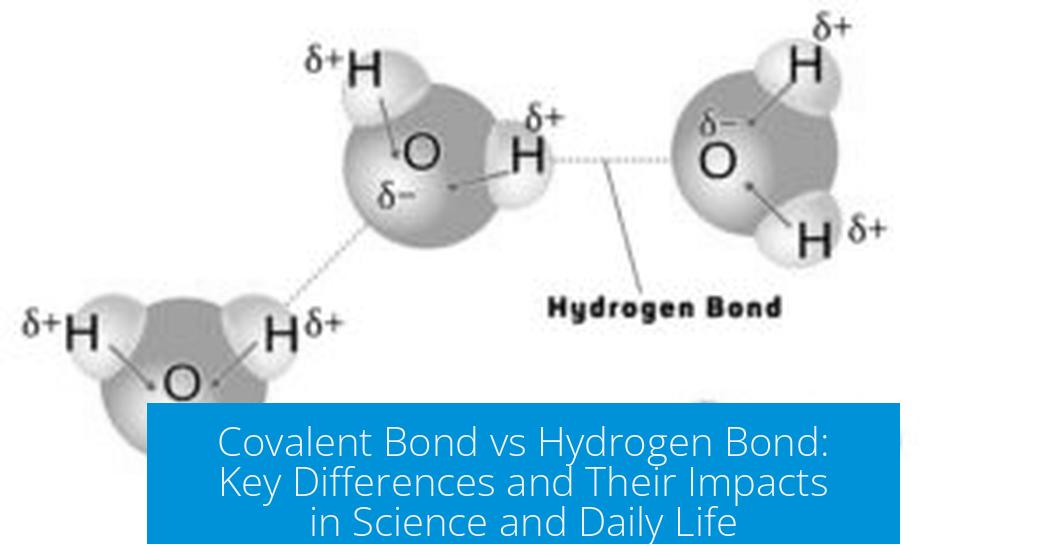
- Water molecule: The oxygen atom attracts electrons, making it partially negative; hydrogen atoms become partially positive. Hydrogen bonds form between water molecules, leading to water’s high boiling point and surface tension.
- DNA double helix: Hydrogen bonds between complementary nitrogenous bases stabilize the double-stranded structure without sharing electrons, allowing strands to separate during replication.
These examples illustrate how hydrogen bonds support biological structures by facilitating molecular recognition and stability while allowing dynamic flexibility.
7. Summary Comparison
| Aspect | Covalent Bond | Hydrogen Bond |
|---|---|---|
| Electron Sharing | Yes, electrons shared to form bond | No, electrostatic attraction between partial charges |
| Bond Strength | Very strong (high energy required to break) | Moderate; weak compared to covalent bonds |
| Bond Formation | Between atoms within molecule | Between molecules or within large molecules |
| Examples | C–H, O–H in methane or water | H–O, H–N, H–F in water, DNA base pairing |
Key Takeaways
- Covalent bonds are strong intramolecular bonds formed by electron sharing between atoms.
- Hydrogen bonds are weaker intermolecular attractions due to polarity and partial charges.
- Electron distribution in covalent bonds creates polarity; hydrogen bonds form from partial opposite charges.
- Hydrogen bonds have biological importance despite being weaker than covalent bonds.
- Bond types form a continuum with overlapping characteristics but differ fundamentally in their electron interactions.
Covalent Bond vs Hydrogen Bond: What’s the Real Difference?
Let’s cut to the chase: The main difference between a covalent bond and a hydrogen bond lies in how atoms connect with each other. Covalent bonds involve atoms sharing electrons, forming a strong, stable connection. Hydrogen bonds don’t share electrons at all—they arise from electrostatic attraction between partially charged atoms.
Confused already? Stick with me. These two bond types shape everything around us, from the water we drink to the very DNA in our cells. Let’s explore what makes them tick, one bond at a time.
1. The Basics: Sharing Electrons vs. Magnet Magic
Think of covalent bonds like welding two bits of metal together. Atoms *share* electrons to fill their outer shells—the famous “octet rule.” They’re so committed that it takes a ton of energy to break these bonds. Seriously, you won’t just boil water to shred its covalent bonds!
Hydrogen bonds? They’re more like two magnets sticking together—not welded, just attracted. The key here is opposite charges pulling on each other. Imagine a lone hydrogen with a slight positive charge getting mysteriously pulled toward a negative partner nearby. This connection is strong for a non-covalent interaction but still much weaker than that welded covalent bond.
2. Why Are Covalent Bonds So Much Stronger?
It boils down to electron sharing. Covalent bonds involve electrons orbiting both nuclei, creating a super stable situation. In water, for instance, oxygen hogs the shared electrons slightly, thanks to its greater electronegativity—making the O-H bond polar but still covalent. Hydrogen, meanwhile, sticks with only one covalent bond. This duo holds tight together.
Hydrogen bonds form because oxygen’s tug on electrons leaves hydrogen looking a little naked—*like a proton exposed to the world*. This positive “naked” spot on hydrogen gets attracted to the lone pairs on oxygen or nitrogen atoms nearby, creating that dipole-dipole attraction we call a hydrogen bond.
3. When Strength Matters: Bond Durability
Want to break covalent bonds? Prepare for a real challenge. These bonds are the foundation stones of molecules. Boiling water only shakes the molecules loose from each other—it doesn’t knock apart the O-H bonds inside each water molecule.
Hydrogen bonds, on the other hand, are more delicate. They’re strong enough to hold water molecules close, which is why water sticks around as a liquid at room temperature instead of evaporating away like methane gas. But heat it up, and these bonds quickly break. The ice melts because hydrogen bonds break, not the covalent ones.
4. A Closer Look: Polarity and Partial Charges
| Bond Type | Electron Sharing | Polarization | Charge Distribution |
|---|---|---|---|
| Covalent Bond | Yes | Polar or Nonpolar | Shared electrons create partial charges if polar |
| Hydrogen Bond | No | Between polar molecules | Partial positive on H, partial negative on O/N/F |
When two atoms share electrons unequally, the bond becomes polarized. Oxygen’s high electronegativity drags electrons closer, leaving hydrogens slightly positive. This setup creates a dipole moment. Hydrogen bonding exploits this polarity by attracting the partially positive hydrogens of one molecule to partially negative atoms of another.
5. Can’t We Just Call It One Bond with Shades?
Here’s a curveball: these bonds aren’t black-and-white categories. Hydrogen bonds blur the line. They’re essentially extreme dipole interactions, leaning on the edge of becoming covalent-ish but not quite crossing that line. So is hydrogen bonding just a “supercharged” dipole force? Many scientists think so, although traditional chemistry classes stick to neat labels.
The takeaway? Don’t overthink the labels. Focus on what the bonds do rather than how they’re categorized. It’s the strength, behavior, and context that matter most.
6. Real-World Examples That Show Their Power
- Water: Oxygen-hydrogen covalent bonds hold the molecule together, while hydrogen bonds connect different water molecules, giving water its unique properties like surface tension and high boiling point.
- DNA: Covalent bonds form the sugar-phosphate backbone, holding strands stable. But hydrogen bonds between nucleotide bases hold the double helix shape together, allowing DNA to unzip during replication—a beautiful balance of strength and flexibility.
7. Why You Should Care: The Impact on Daily Life and Science
Hydrogen bonding explains why ice floats (it’s less dense than water because hydrogen bonds hold molecules further apart in solid form). It’s also why proteins fold the way they do. Without hydrogen bonds, life as we know it wouldn’t exist. Covalent bonds, meanwhile, are the foundation of all molecules—without them, no stable matter.
8. Summing It Up With a Handy Comparison
| Feature | Covalent Bond | Hydrogen Bond |
|---|---|---|
| Electron Sharing | Yes, mutual sharing fills outer shells | No electron sharing; electrostatic attraction only |
| Bond Strength | Very strong (requires lots of energy to break) | Moderate strength, noticeable but easily broken by heat |
| Bond Examples | C-H in methane, O-H in water molecule | H bonded to N, O, F (water molecules, DNA bases) |
| Role in Molecules | Primary structure, molecular framework | Secondary interactions, molecule stabilization |
9. Quick Tips for Students and Curious Minds
- If you remember one thing: covalent bonds share electrons, hydrogen bonds don’t.
- Think of covalent bonds as permanent connections and hydrogen bonds more as “friendly handshakes.”
- Looking at water? The oxygen-hydrogen bond inside a molecule is covalent; connections between molecules are hydrogen bonds.
- Always remember the electrostatic nature of hydrogen bonds—this link relies on polarity, not electron sharing.
10. Final Thought: More Than Just Chemistry Terms
Understanding these bonds is like cracking the code for life’s molecular dance. While textbooks may lead you to see covalent and hydrogen bonding as separate worlds, the reality is a continuum with shades and overlaps.
So next time you sip water or marvel over DNA’s intricate structure, remember: it’s these bonds—one strong and welded, the other a magnetic attraction—that make it all possible. And isn’t that a humbling mix of chemistry and magic?
“Chemistry is not just about elements and bonds. It’s about connections—some strong like covalent welds, others gentle but vital like hydrogen’s magnetic handshake.”
Want to dive deeper? Check out resources like the LibreTexts Chemistry Library for hands-on explanations and examples.
What is the main difference between covalent and hydrogen bonds?
Covalent bonds involve the sharing of electrons between atoms. Hydrogen bonds form due to electrostatic attraction between a partially positive hydrogen and a electronegative atom, without electron sharing.
How does the strength of covalent bonds compare to hydrogen bonds?
Covalent bonds are very strong and hold atoms firmly together. Hydrogen bonds are weaker and can be broken by moderate heat or movement but are strong enough to influence molecular behavior like water’s liquid state.
Can hydrogen bonds be considered a type of covalent bond?
Hydrogen bonds are not true covalent bonds since electrons are not shared. However, they represent a strong dipole interaction and sometimes have covalent-like features but remain distinct in nature.
Why are hydrogen bonds important in biological molecules like DNA?
Hydrogen bonds hold DNA strands together by connecting base pairs. These bonds provide stability while allowing strands to separate for replication, unlike the stronger covalent bonds within each strand.
Do hydrogen bonds involve electron sharing like covalent bonds?
No, hydrogen bonds do not involve sharing electrons. Instead, partial charges attract one another, creating a bridge between molecules without changing their internal covalent bonds.




Leave a Comment