How to Differentiate Between Ionic and Covalent Bonds
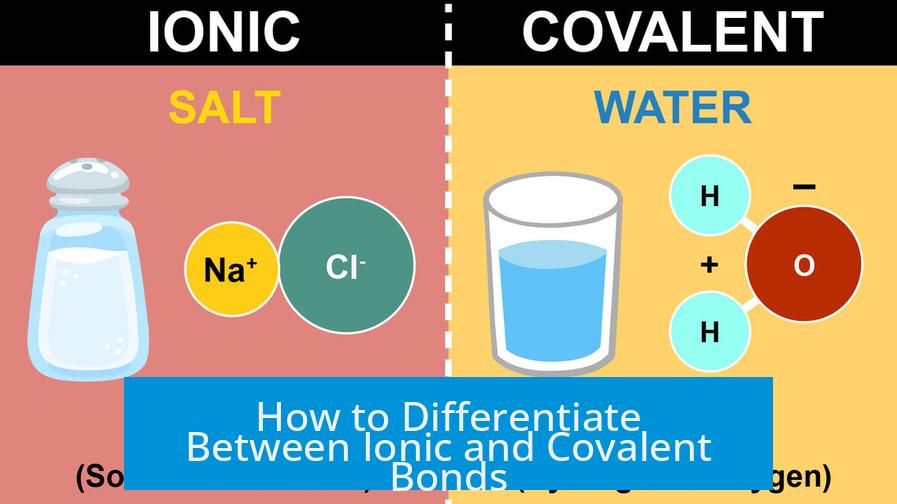
At its core, differentiating ionic from covalent bonds involves understanding how electrons are distributed between atoms. Ionic bonds form when electrons transfer from one atom to another, resulting in charged ions held together by electrostatic forces. Covalent bonds arise when atoms share electrons, either equally or unequally. However, this distinction is not always clear-cut, as bonding lies on a spectrum from purely covalent to primarily ionic.
The Bonding Spectrum: From Covalent to Ionic
Chemical bonds do not fall neatly into two categories. Instead, they span a continuum based on how electrons are shared or transferred. At one end, purely covalent bonds involve equal sharing of electrons between atoms. At the other extreme, ionic bonds represent an almost complete electron transfer, creating distinct positive and negative ions.
Between these extremes are polar covalent bonds. These involve unequal sharing of electrons, with one atom attracting them more strongly than the other. The greater the difference in this attraction, the more polar the bond becomes, edging closer to ionic character.
“Imagine sharing a jointly owned toy with your brother. If he rarely lets you play with it, at what point do you consider it stolen rather than shared?”
This analogy expresses the challenge in defining the exact point where polar covalent bonds become ionic. Instead of strict categories, chemists often describe bonds by their ionic character, a measure of how much electron transfer occurs.
Electronegativity Differences and Limitations
One common method to differentiate bonds involves comparing the electronegativities of involved atoms. Electronegativity is an atom’s ability to attract electrons. The larger the difference in electronegativity (ΔEN), the more ionic the bond is assumed to be.
| Electronegativity Difference (ΔEN) | Expected Bond Type |
|---|---|
| 0 – 0.4 | Nonpolar Covalent |
| 0.4 – 1.7 | Polar Covalent |
| Greater than 1.7 | Predominantly Ionic |
However, this cutoff is not absolute. For example:
- Aluminum bromide (AlBr3) has ΔEN around 1.3, near the borderline, but behaves covalently due to its molecular structure.
- Beryllium chloride (BeCl2) has ΔEN near 1.5 but forms polymeric covalent structures rather than classic ionic lattices.
Such examples illustrate why electronegativity difference alone cannot fully determine bond type. Context and other factors must be considered.
Fajan’s Rules and Covalent Character in Ionic Bonds
Fajan’s rules help explain why some bonds classified as ionic show significant covalent character. The rules identify factors increasing the covalent nature in otherwise ionic bonds:
- Ion size: Smaller cations and larger anions increase covalent character by enhancing polarization.
- Cation charge: Higher positive charge on the cation intensifies polarization of the anion, increasing covalent character.
Polarization occurs when the positive ion distorts the electron cloud of the negative ion, leading to electron sharing tendencies. As a result, some ionic bonds gain partial covalent behavior.
This means aluminum bromide and beryllium chloride are better viewed as covalent or covalent-like rather than truly ionic, due to small cation size and charge-related polarization effects.
Physical and Experimental Differences
Physical properties and experimental evidence provide further clues to bond type:
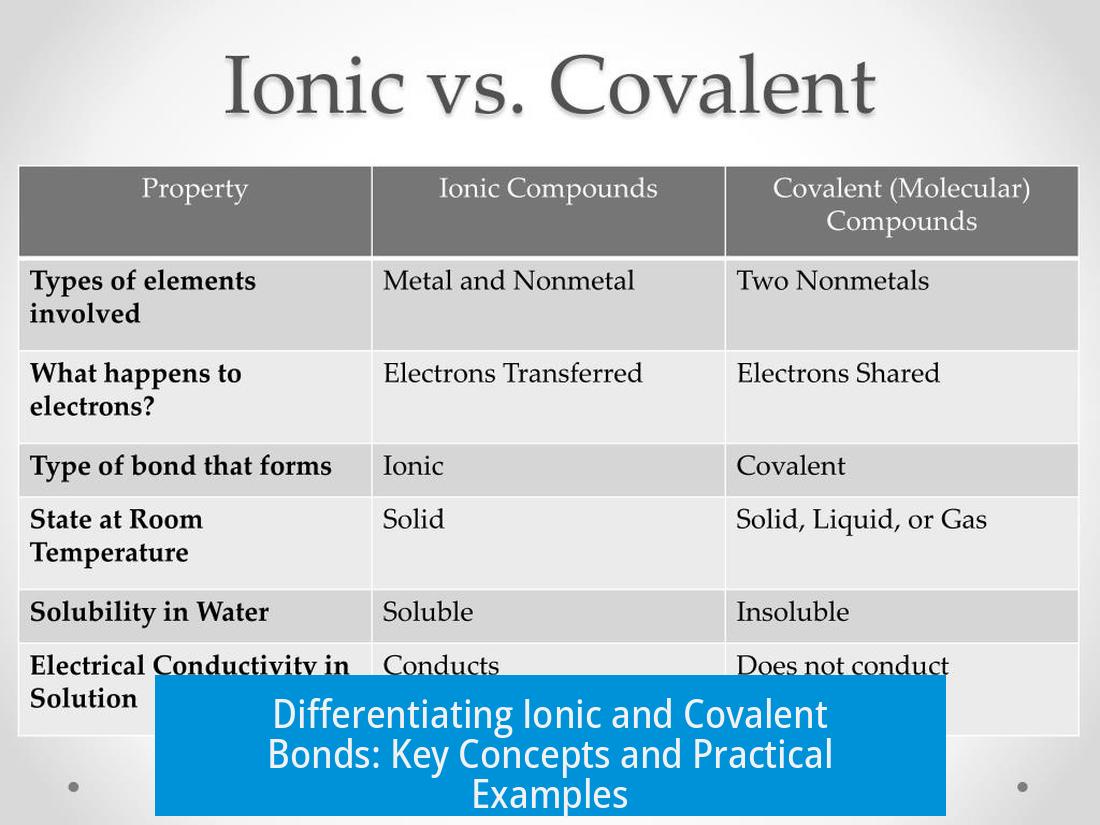
- Structure: Ionic compounds often form large, repeating lattices rather than discrete molecules, as seen in sodium chloride (NaCl).
- Nuclear Magnetic Resonance (NMR): Covalent molecular bonds can show through-bond nuclear coupling, which is absent or weak in ionic interactions.
- Solubility and Melting Points: Ionic compounds tend to have high melting points and dissolve readily in polar solvents like water.
However, some covalent substances, such as phosphorus allotropes, challenge these distinctions due to their complex structures.
Common Misconceptions and Practical Advice
A common misconception is that ionic bonds always involve complete electron transfer, and covalent bonds always involve equal sharing. Reality shows a gradient of bonding behaviors.
Practical tips for differentiation:
- Assess electronegativity differences but recognize they provide guidelines, not strict rules.
- Consider the metal/nonmetal nature of elements: bonds between metals and nonmetals tend to be more ionic.
- Use Fajan’s rules to analyze ion size and charge effects.
- Observe physical properties and molecular structures where possible.
- Use the concept of ionic character to describe a bond’s nature rather than assign discrete categories.
Examples Illustrating Nuances
Even well-studied bonds defy clear classification:
- Organometallic complexes exhibit bonds with partial ionic and covalent features, complicating simple definitions.
- Hydrogen bonding itself blurs lines between covalent and noncovalent interactions.
- Some compounds traditionally called ionic possess significant covalent character due to ion polarization effects.
Chemistry teaches that bonding is nuanced, and rigid separation into ionic vs. covalent often oversimplifies the true nature of atomic interactions.
Key Takeaways
- Bonds exist on a spectrum from purely covalent (equal sharing) to primarily ionic (electron transfer).
- Electronegativity difference guides bond type but has no absolute cutoff.
- Fajan’s rules explain why some ionic bonds show covalent character due to ion size and charge.
- Physical structure and experimental evidence help differentiate bonds but are not always definitive.
- Describing bonds by their ionic character is more accurate than strict ionic/covalent labels.
- Bonding theories reflect chemical complexity and should be applied flexibly.
What is the main difference between ionic and covalent bonds?
Ionic bonds form when electrons transfer fully from one atom to another, creating charged ions. Covalent bonds involve atoms sharing electrons more or less equally, forming molecules.
How can electronegativity difference help distinguish between ionic and covalent bonds?
Electronegativity difference gives a rough idea: large differences suggest ionic bonds, smaller ones suggest covalent. But no strict cutoff exists, so it’s only a guideline, not a rule.
Why do some ionic bonds show covalent character?
Fajan’s rules explain that small, highly charged cations can distort large anions, making bonds partly covalent. So, many bonds lie on a spectrum rather than being purely ionic or covalent.
Can physical properties indicate whether a bond is ionic or covalent?
Yes. Ionic compounds often form lattice structures with repeating charged ions, while covalent compounds form discrete molecules. Testing techniques like NMR can also give clues about bonding types.
Is it always clear-cut to classify a bond as ionic or covalent?
No, many bonds sit between these extremes. Considering “ionic character” helps understand bonding without strict categories. This view reflects the chemical bonding continuum more realistically.



Leave a Comment