Understanding Phosphorylation–Dephosphorylation Enzymatic Reactions
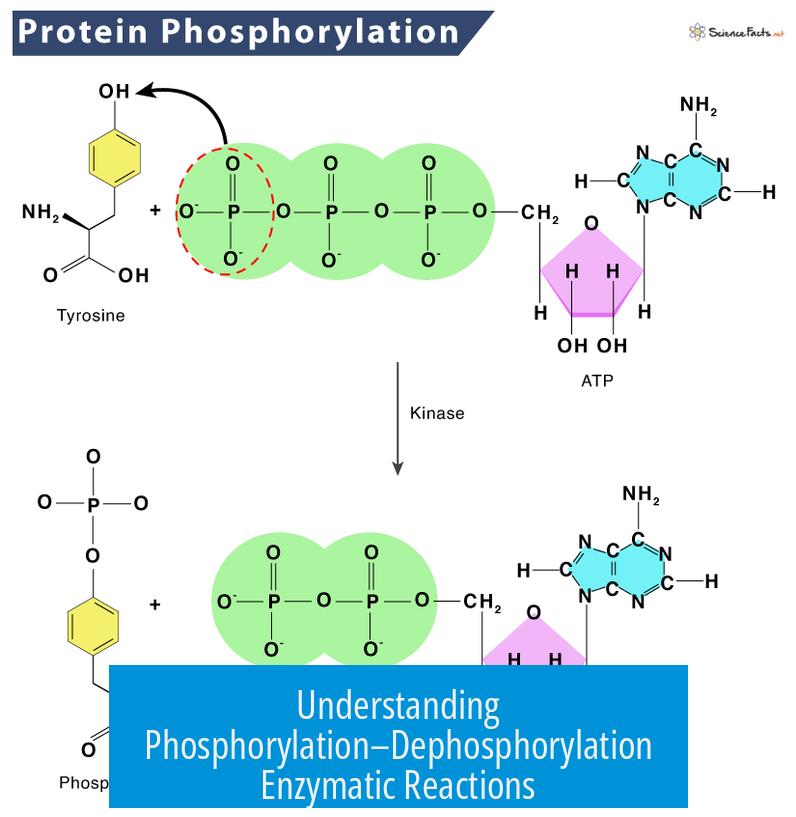
Phosphorylation–dephosphorylation enzymatic reactions involve the addition or removal of a phosphate group to or from a molecule, typically mediated by enzymes. These reactions regulate many cellular processes by switching enzymes or proteins “on” or “off.”
What is Phosphorylation?
Phosphorylation means adding a phosphate group (PO43−) to a molecule. This is often done using a phosphate donor such as ATP (adenosine triphosphate). For example, ADP (adenosine diphosphate) can gain a phosphate group and become ATP. This process stores energy within the molecule.
What is Dephosphorylation?
Dephosphorylation is the removal of a phosphate group from a molecule. When ATP loses a phosphate to become ADP, it releases energy. Cells harness this energy to power many biological functions like muscle contraction or biochemical reactions.
Role of Enzymes in Phosphorylation and Dephosphorylation
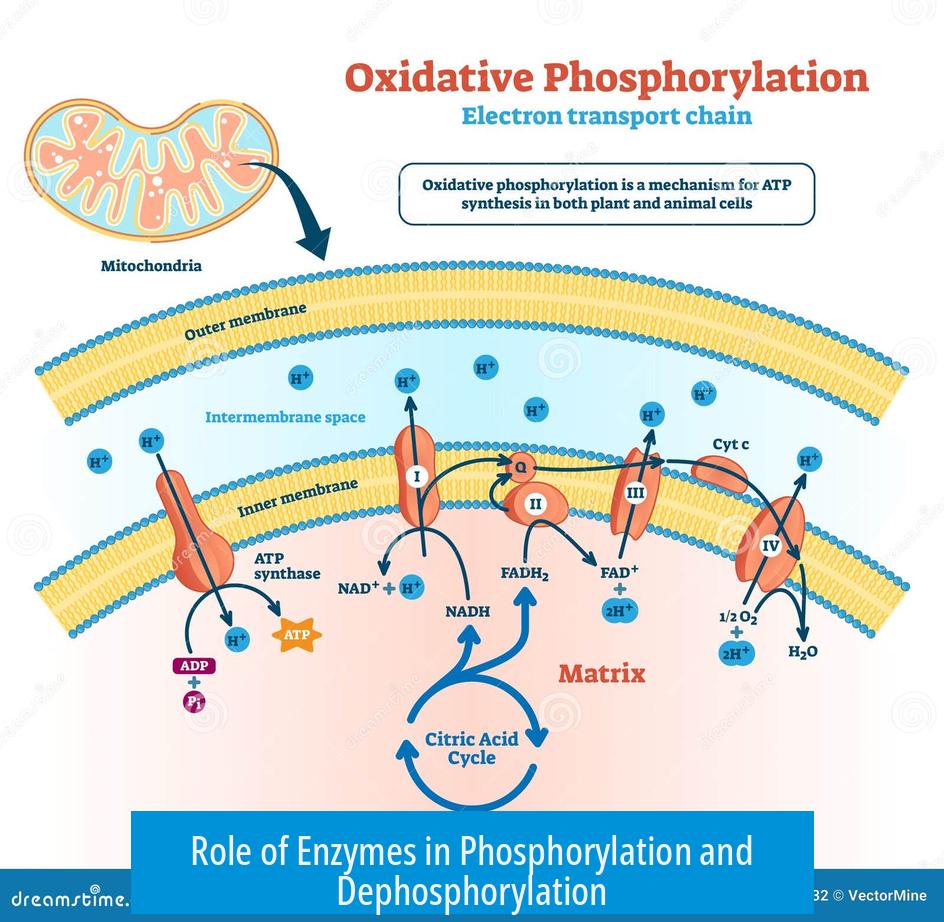
- Specific enzymes add phosphate groups, called kinases.
- Other enzymes remove phosphate groups, called phosphatases.
- By switching enzymes between phosphorylated and dephosphorylated forms, cells regulate enzyme activity and signal pathways.
Biological Significance
This cycle controls vital processes. For instance, phosphorylation can activate an enzyme to speed up a reaction. Dephosphorylation can inactivate it. Energy transfer in cells often involves these reactions, such as turning ADP into ATP or vice versa.
Example: Pyruvate Kinase Reaction
Pyruvate kinase transfers a phosphate group between phosphoenolpyruvate (PEP) and ADP. In living cells, it usually works one way, producing ATP. In laboratory conditions, this reaction can be reversed by adjusting concentrations of reactants and products.
| Reaction | Direction in Cells | Direction in Test Tube |
|---|---|---|
| PEP + ADP ⇌ Pyruvic acid + ATP | Forward (ATP production) | Both (reversible by concentration) |
Energy Changes and Reaction Direction
These reactions often have a large free energy change. This means the reaction strongly favors one direction under normal cellular conditions, preventing it from easily reversing. This directionality is essential for controlled energy flow in the cell.
Key Takeaways
- Phosphorylation adds phosphate groups; dephosphorylation removes them.
- Enzymes regulate these changes to control cellular functions.
- ATP is a common phosphate donor and energy carrier.
- These reactions release or store energy essential for life.
- Some reactions are reversible but direction depends on conditions.
What does phosphorylation mean in simple terms?
Phosphorylation means adding a phosphate group to a molecule. This small change can change how the molecule works.
Why is dephosphorylation important in cells?
Dephosphorylation removes a phosphate group. It often switches off or changes the function of molecules in cells, helping control cell activities.
How do enzymes change during phosphorylation and dephosphorylation?
Enzymes can be turned on or off by adding or removing phosphate groups. This helps regulate their activity in the cell.
What role does ATP play in phosphorylation?
ATP stores energy by holding phosphate groups. When a phosphate group is removed from ATP, energy is released for cell processes.
Are phosphorylation and dephosphorylation reactions reversible?
Some reactions can go both ways, like the one involving pyruvate kinase. However, many move mostly in one direction due to energy changes.





Leave a Comment