Reduction with DIBAL-H: A Detailed Overview
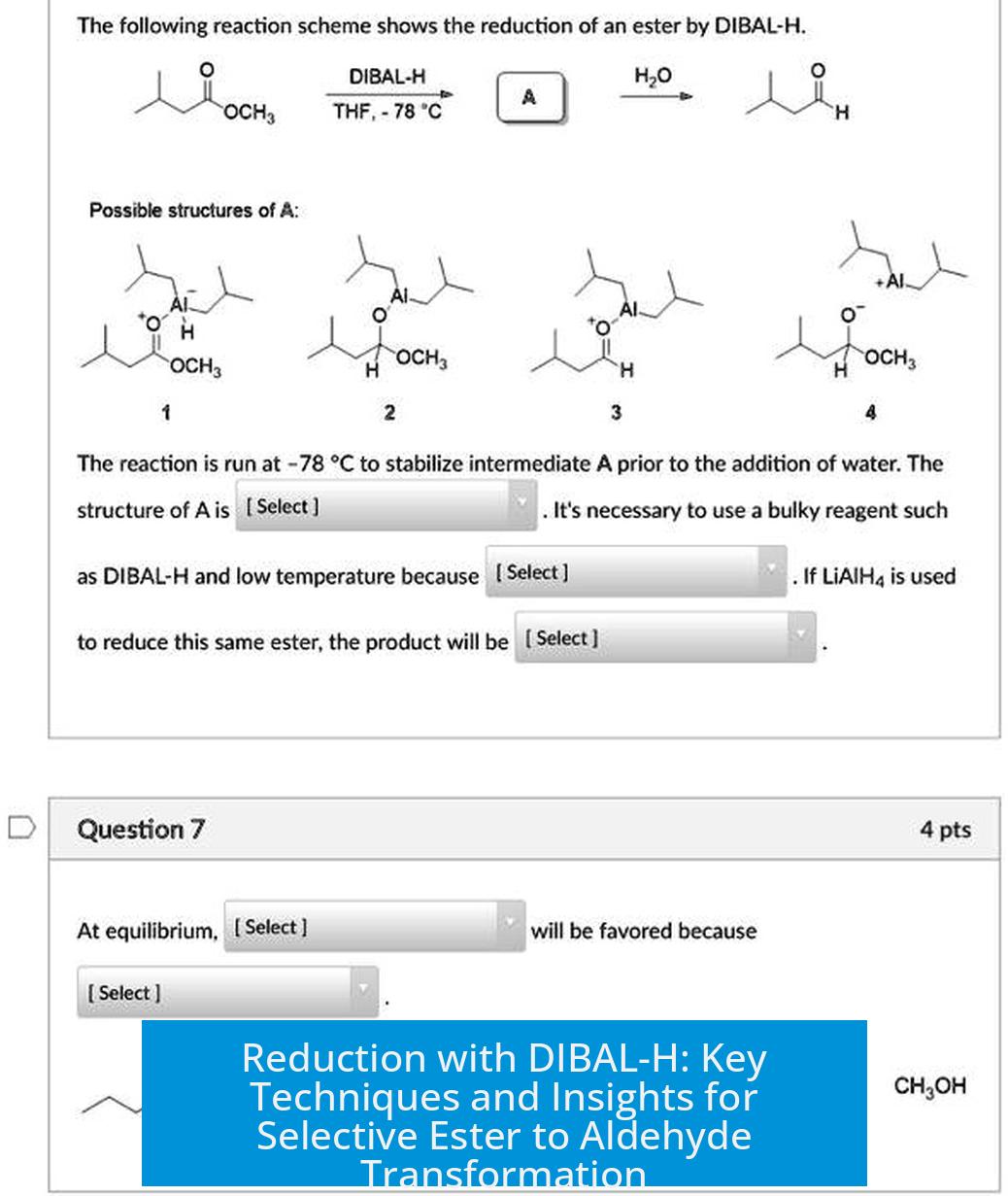
DIBAL-H reduction of esters to aldehydes requires careful control due to the exothermic nature and fast kinetics of the reaction. This process involves converting esters into aldehydes selectively, but practical challenges often arise.
Exothermicity and Temperature Control
DIBAL-H reductions release significant heat locally despite the overall reaction mixture being cooled to -78°C. Each added drop of DIBAL heats the solution momentarily, increasing the local temperature near the ester substrate.
This local warming can accelerate the reaction and affect the stability of intermediates.
Reaction Kinetics and Intermediate Stability
The reaction proceeds quickly, forming a tetrahedral intermediate. Because of ongoing temperature fluctuations caused by DIBAL addition, this intermediate easily proceeds to collapse into the aldehyde.
The aldehyde thus formed is more reactive and prone to further reduction to the alcohol if conditions are uncontrolled.
Challenges in Selective Reduction
- Selective reduction stopping precisely at the aldehyde stage is difficult due to the aldehyde’s higher reactivity.
- In practice, many chemists opt to reduce esters fully to alcohols and then oxidize back to aldehydes to improve yields and selectivity.
Special Case: Lactones
Lactones behave differently under DIBAL reduction, owing to their cyclic structure. They form stable tetrahedral intermediates known as cyclic hemiacetals.
These intermediates generally do not proceed to overreduction, making lactone reductions more controlled.
Best Practices for Reducing Esters to Aldehydes
- Lower the reaction temperature further to around -100°C to curb excess reaction speed.
- Submerge a large part of the flask into a cooling bath for better heat dissipation.
- Add DIBAL slowly as a dilute solution in solvents like hexane or toluene, preferably on the side of the flask.
- Ensure each aliquot cools before contacting the ester to minimize local heating.
These steps help maintain selectivity and prevent uncontrolled overreduction to alcohols.
Key Takeaways
- DIBAL reductions are highly exothermic with rapid kinetics requiring precise temperature control.
- Tetrahedral intermediates easily collapse to aldehydes, which can be further reduced.
- Selective ester-to-aldehyde reduction is challenging; often alcohol formation followed by oxidation is preferred.
- Lactones form stable intermediates that resist overreduction.
- Slow addition at very low temperatures using diluted solutions maximizes control over the reaction.
How does the exothermic nature of DIBAL-H affect ester reductions?
DIBAL-H reductions release heat locally with each drop added. Even if the reaction mixture is cooled to -78 °C, local heating can raise temperatures where dibal contacts the ester.
Why is selective reduction of esters to aldehydes with DIBAL challenging?
The initial tetrahedral intermediate formed is unstable and can collapse to a more reactive aldehyde. This aldehyde may be further reduced to an alcohol, making selective reduction difficult.
How can one improve the selectivity of ester reduction to aldehyde using DIBAL?
- Cool the reaction below -78 °C, near -100 °C.
- Use a diluted dibal solution in hexane or toluene.
- Add dibal slowly at the side of the flask for better cooling.
- Immerse the flask deeply in the cooling bath.
Why don’t lactones overreduce under DIBAL reduction like esters do?
Lactones form stable cyclic hemiacetal intermediates when reduced. These intermediates resist further reduction, preventing overreduction to alcohols.




Leave a Comment