Understanding H2O Bent Molecular Geometry
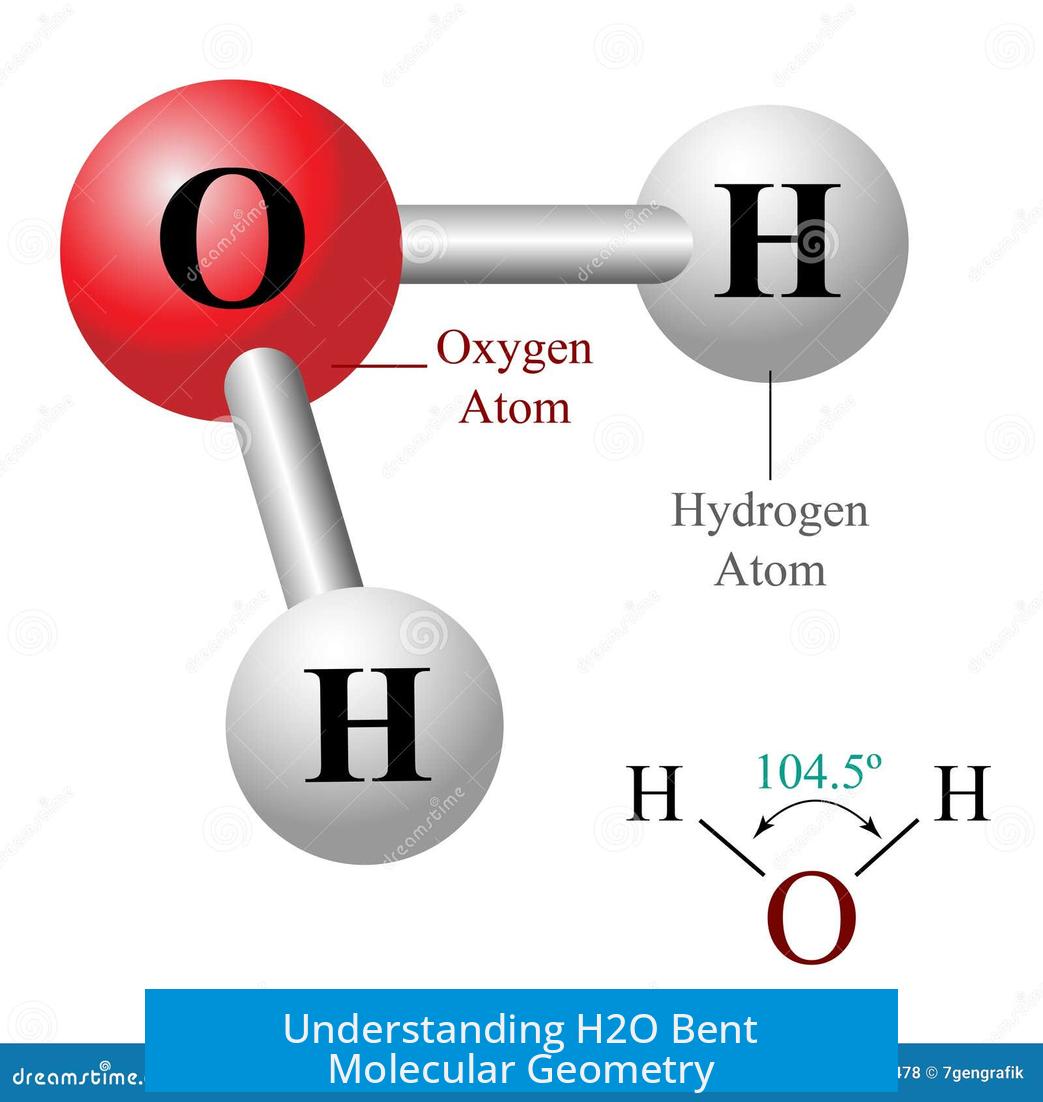
Water (H2O) exhibits a bent molecular geometry due to the arrangement of its two bonded hydrogen atoms and two lone pairs of electrons on oxygen, resulting from electron repulsion and spatial distribution that deviates from an ideal tetrahedral shape. This distortion influences the bond angles, electron density, and overall molecular symmetry.
Molecular Shape and Lone Pairs
Water’s shape can be visualized as a distorted tetrahedron. Oxygen at the center bonds with two hydrogen atoms and hosts two lone pairs of electrons. These lone pairs occupy space but are not visible as atoms.
The lone pairs’ electron density is arranged out of the plane formed by the hydrogen atoms. This spatial positioning alters the molecular geometry from linear or perfectly tetrahedral to bent. Lone pairs repel more strongly than bonding pairs, influencing the shape significantly.
3D Spatial Arrangement and Lone Pair Repulsion
In three dimensions, water mimics a tetrahedral electron pair geometry where all four electron pairs (two bonding and two lone pairs) aim to maximize their distance from each other to minimize repulsion. However, the lone pairs force the hydrogen atoms closer together by bending the molecular structure.
The lone pairs position themselves to be as far apart as possible, bending out of the plane, which isn’t always evident in two-dimensional drawings. This arrangement explains water’s distinct bent shape rather than a flat, symmetrical shape.
Bond Angle Differences Between Methane and Water

| Molecule | Bond Angle (°) |
|---|---|
| Methane (CH4) | 109.5 |
| Water (H2O) | 105 |
Methane, with four bonded atoms and no lone pairs, maintains a perfect tetrahedral bond angle of 109.5°. Water’s bond angle shrinks to approximately 105° because lone pairs repel more strongly than bonding pairs. This repulsion pushes the hydrogen atoms together, creating the bent shape.
Limitations of the Lone Pair Model and Role of Molecular Orbital Theory
Traditional lone pair models effectively describe water’s shape but have limitations. They do not fully explain the underlying quantum mechanical reasons for the molecular geometry or bonding characteristics.
Molecular Orbital (MO) theory provides a deeper explanation. It accounts for electron delocalization and quantum interactions, offering a precise description of water’s bent shape based on electronic structure rather than just electron pair repulsions.
Molecular Orbital Diagram and Molecular Symmetry
The MO diagram for water details the energy levels and spatial characteristics of molecular orbitals, showing how the electrons fill these orbitals to stabilize the molecule.
Water exhibits C2v molecular symmetry in its ground state. This symmetry governs how the orbitals overlap and interact and also supports the bent shape by defining allowed molecular orbitals and their orientation in space.
Details about the MO diagram and symmetry character table help explain why water does not adopt a planar or linear shape but settles into a bent geometry consistent with minimum energy and symmetry constraints.
Summary of Model Accuracy and Molecular Geometry Insights
- Lone pair models offer practical insight but cannot fully explain water’s molecular geometry.
- The bent geometry arises from repulsion differences: lone pairs repel more than bonding pairs.
- Molecular Orbital theory offers a quantum-level understanding of the electron distribution and molecular symmetry.
- Water’s bond angle is about 105°, compressed from the tetrahedral ideal of 109.5° due to lone pair effects.
- The combination of electron repulsion and molecular symmetry determines the stable bent shape.
Key Takeaways
- Water’s bent geometry stems from having two lone pairs and two bonded hydrogen atoms around oxygen.
- Lone pairs occupy space and repel bonded atoms, reducing bond angles from 109.5° to ~105°.
- The spatial arrangement of electron pairs forms a near-tetrahedral electron pair geometry but a bent molecular shape.
- Molecular Orbital theory provides a more precise, quantum mechanical explanation than lone pair models.
- Water has C2v symmetry reflecting its bent molecular structure and electronic configuration.
What causes the bent shape of the H2O molecule?
Water’s shape is bent because it has two lone pairs on oxygen. These lone pairs occupy space and repel the hydrogen atoms, pushing them closer together. This forms a distorted tetrahedral arrangement with a bond angle near 105°.
Why is the bond angle in water smaller than in methane?
Methane has a 109.5° angle because all four positions are occupied by bonded atoms. In water, lone pairs repel more strongly than bonded atoms, reducing the H–O–H angle to about 105°. Lone pair repulsion changes the geometry.
How do lone pairs influence water’s 3D shape?
Lone pairs sit out of the plane formed by hydrogen atoms and push them closer together to minimize repulsion. In three dimensions, lone pairs and bonds arrange themselves tetrahedrally, but with different angles due to stronger lone pair repulsion.
What does molecular orbital theory say about water’s geometry?
Molecular orbital theory explains water’s bent shape using quantum mechanics. It shows water has C2v molecular symmetry and an electronic structure that causes the bond angle and shape, going beyond the simple lone pair model.
Why are lone pair models insufficient to fully explain water’s shape?
Lone pair models describe geometry simply but do not capture the electronic structure accurately. They cannot explain why lone pairs cause bending at the quantum level, which is clarified by more advanced molecular orbital theory.




Leave a Comment